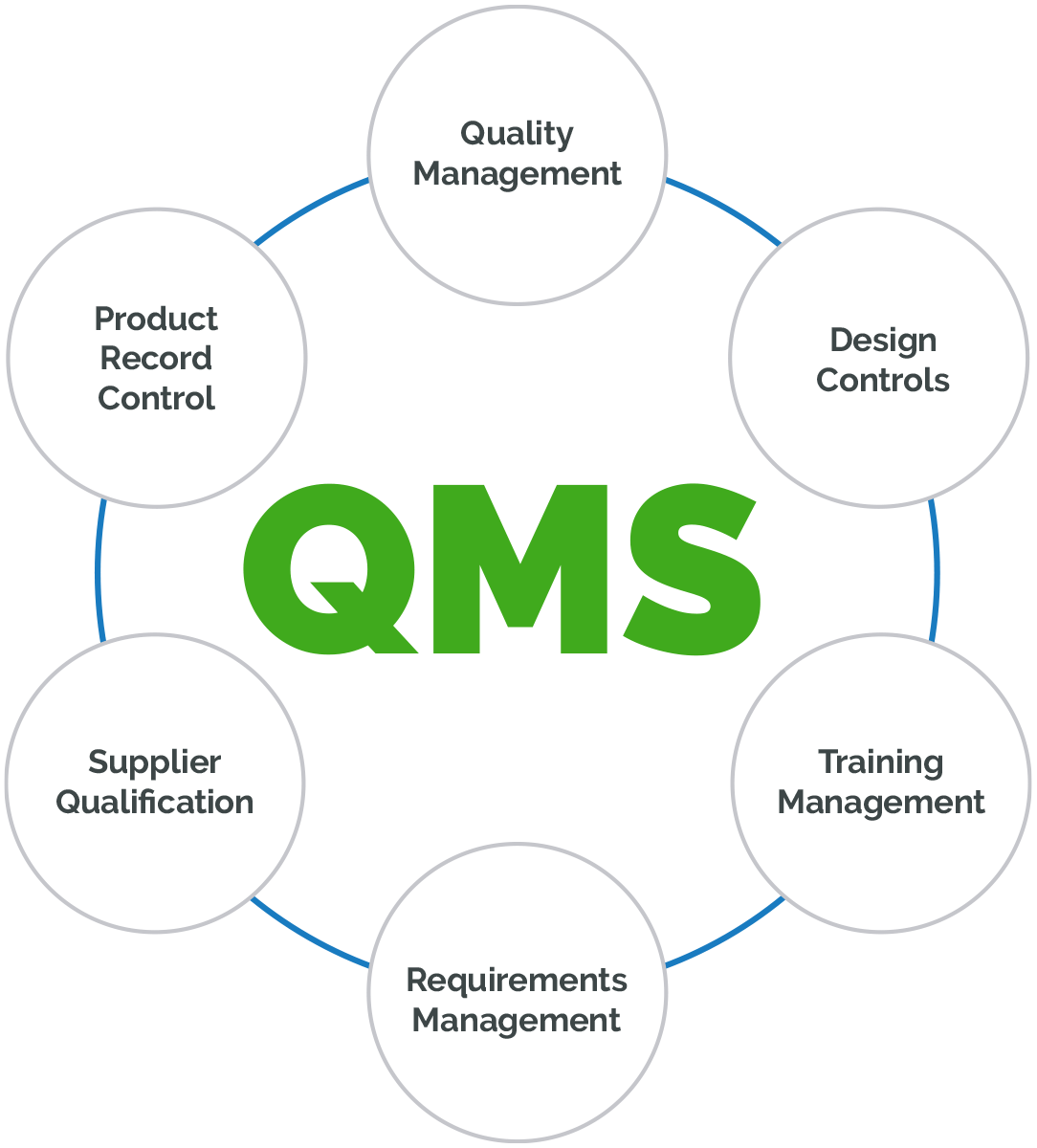
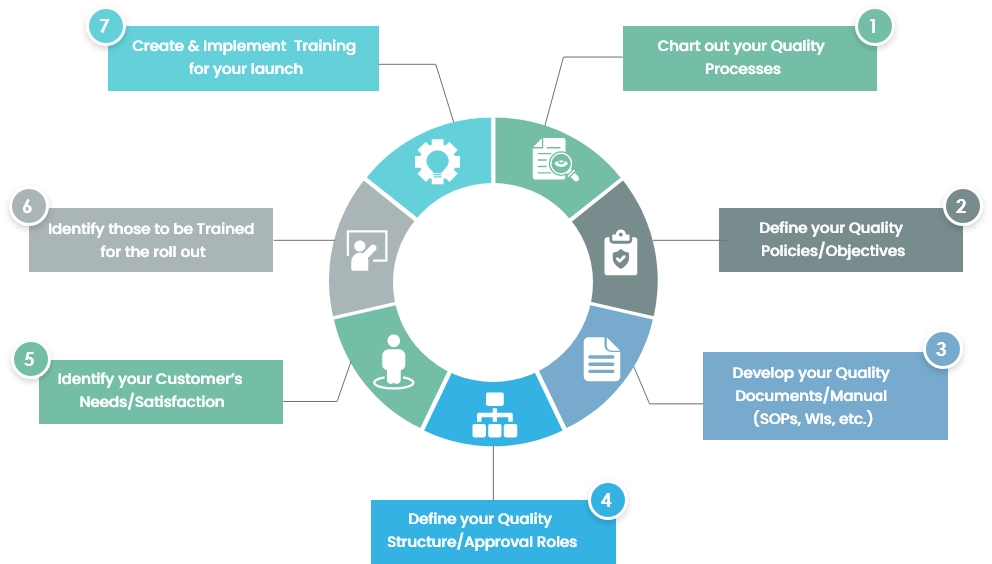
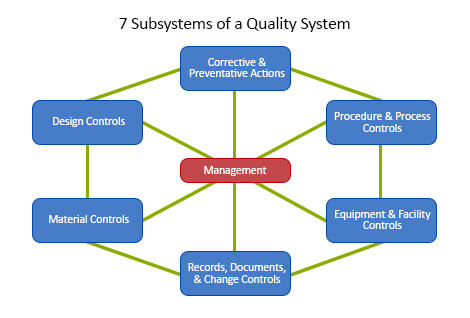
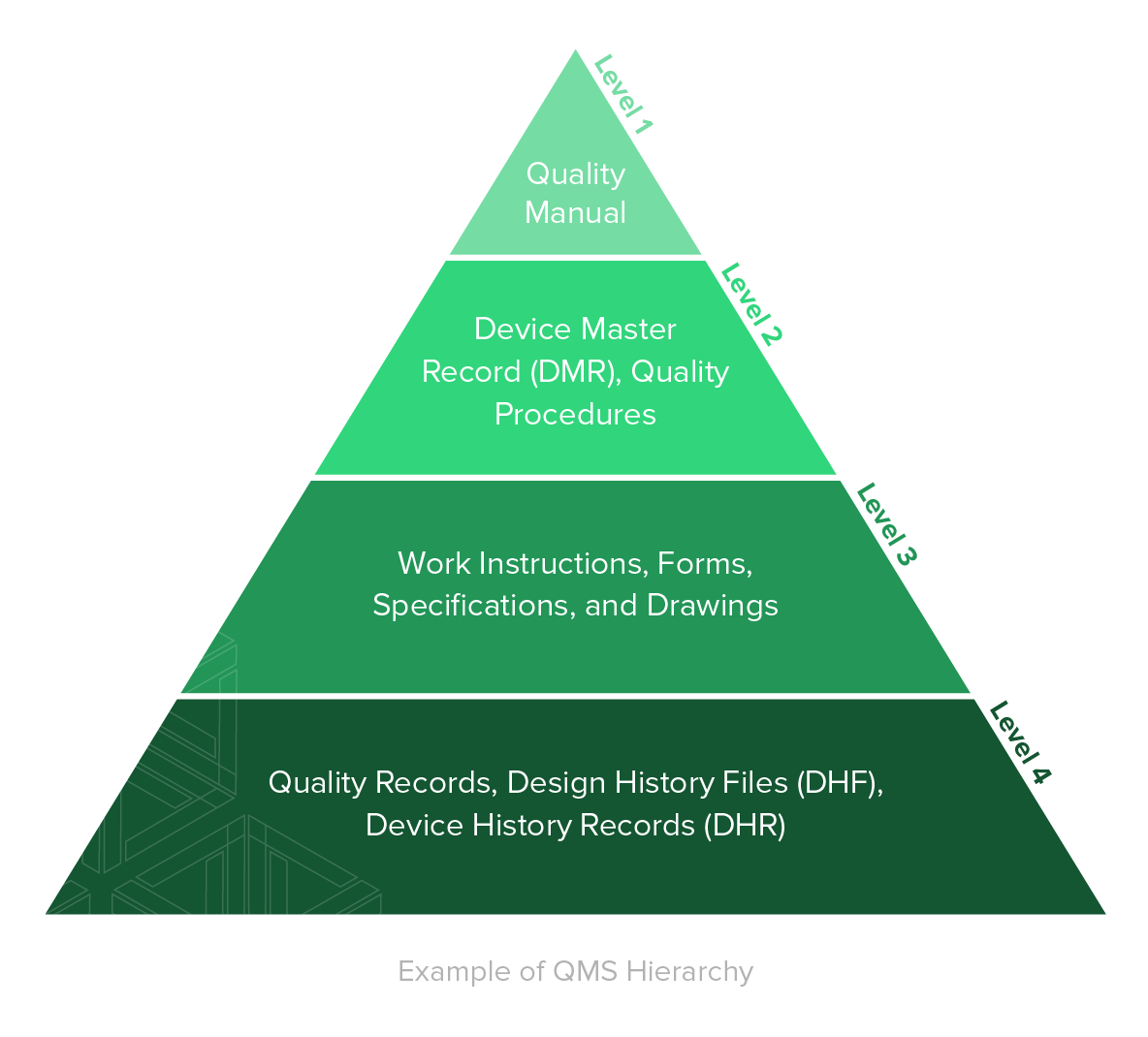
FDA 510(k) Consulting Service for Medical Devices » ISO 13485 Quality Management System Certification

End-to-End Biologics CDMO Quality Management System|OPM Biosciences – Cell Culture Media and End-to-End CDMO

Designing A World-Class Quality Management System For FDA Regulated Industries: Quality System Requirements (QSR) For cGMP : Muchemu, David: Amazon.fr: Livres

Live from PDA/FDA: FDA Considers Incentivizing Quality Management Maturity (QMM) | Healthcare Packaging